Information about HNF4
Overview
Hepatocyte nuclear factor 4 (HNF4, HNF-4) was originally identified as an activity in crude rat liver nuclear extracts that bound DNA elements required for the transcription of two liver-specific genes -- transthyretin (TTR) and apolipoprotein C3 (APOC3). After protein purification and cloning, it was found that HNF4 is a member of the nuclear receptor superfamily (NR2A1) of ligand-dependent transcription factors (Sladek et al., 1990). HNF4 is one of the most highly conserved nuclear receptors and is found in every animal organism examined thus far, from sponge to man (Fig. 1). (Sladek, 2010)
Figure 1. Shown are percent amino acid identity among the various functional domains of the HNF4a protein. Numbers refer to amino acid residues. DBD, DNA binding domain. LBD, ligand binding domain.
Classification of HNF4 as a Novel Nuclear Receptor
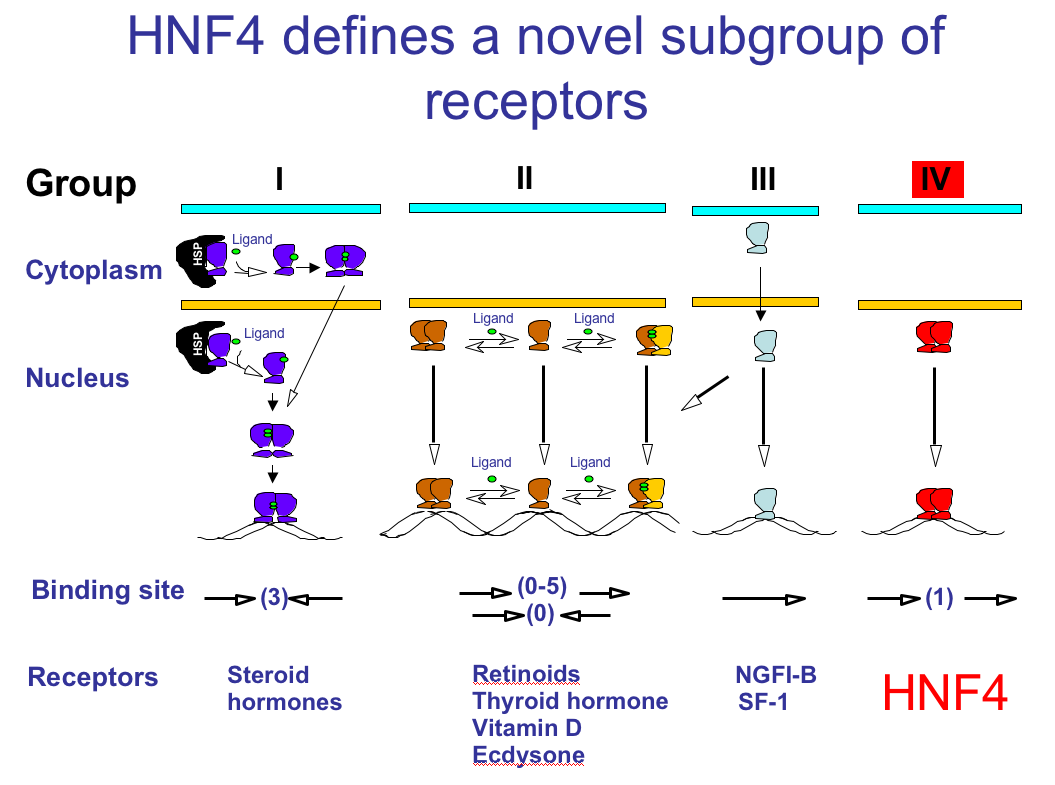
HNF4 is rather unique in that it binds DNA exclusively as a homodimer and yet is localized primarily in the nucleus and binds only DNA response elements consisting of direct repeats (Fig. 2). It also activates transcription and binds several different co-activators (such as GRIP1, SRC, p300/CBP, DRIP205, PGC1) in the absence of exogenously added ligand. It took more than 20 years and many attempts to identify the endogenous ligand for HNF4. We now know that mammalian HNF4alpha expressed under physiological conditions binds a single, essential fatty acid, linoleic acid (LA) (C18:2). However, the exact function of that ligand remains to be determined (Yuan et al., 2009). HNF4 proteins also possess a pair of charged residues in helices 9 and 10 in the ligand binding domain (LBD) that appear to be critical for homodimerization and to play a role in preventing heterodimerization with other receptors such as RXR (Bogan et al., 2000) (Fig. 4).
Figure 2. Nuclear Receptor Subfamilies. (Jiang et al.,1995)
Since HNF4 forms exclusively homodimers, is located only in the nucleus and binds DNA elements consisting of direct repeats, it defines a unique subfamily of nuclear receptors. This suggests that the mechanism of action of HNF4 is distinct from that of other receptors, which definitely appears to be the case. Adapted from Jiang et al., 1995, MCB 15: 5131-5143.
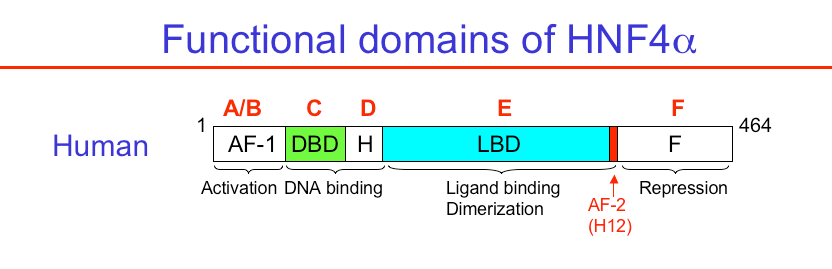
Figure 3. Domain structure of HNF4alpha
Figure 4. Alignment of the amino acids in the region of helix 9 and 10 of all identified HNF4 genes from the species indicated. Dash, amino acids identical to human. The C. elegans HNF4-like protein identified in 1994 does not contain the signature K(X)26E/D motif.
HNF4alpha Gene and Splicing Variations
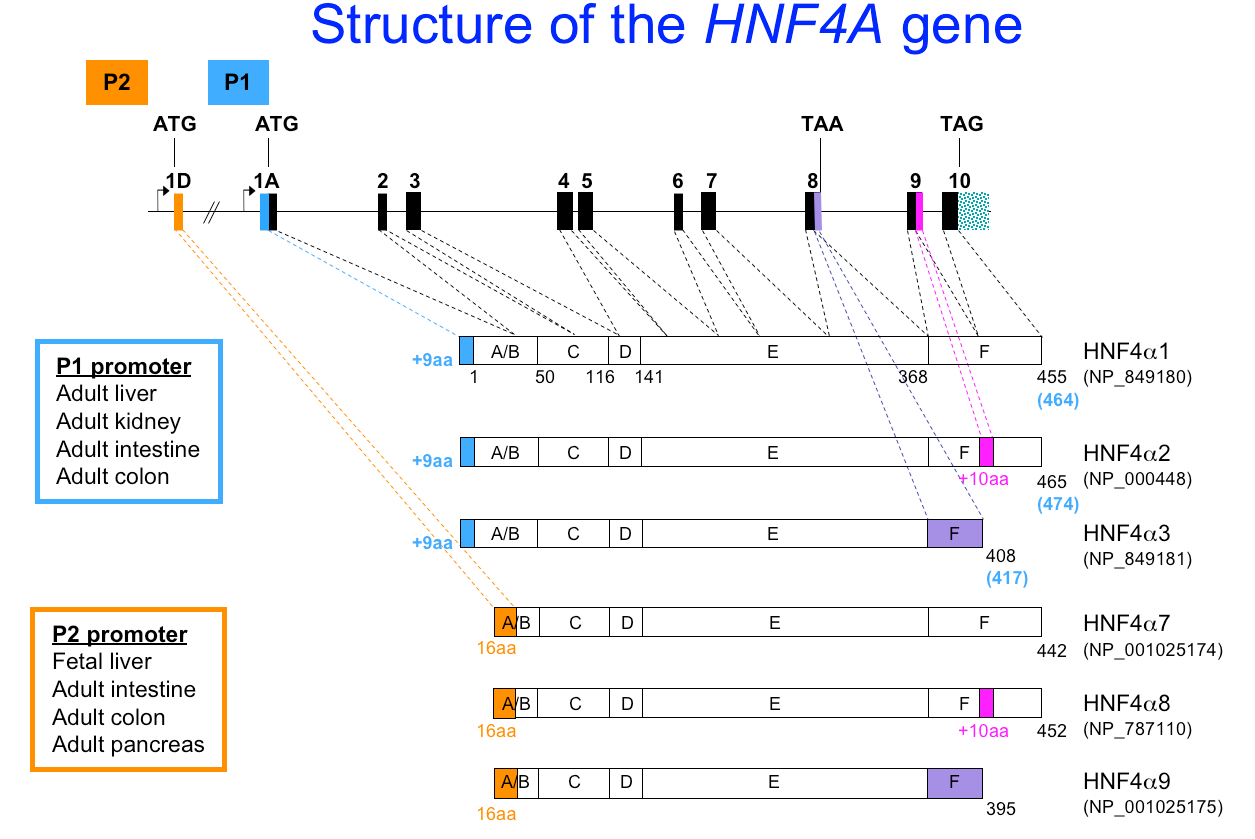
The first HNF4 gene identified is HNF4A (HNF4alpha) and is located on human chromosome 20 q13.1-13.2. The gene consists of at least 12 exons, has two promoters (P1 and P2) and spans >60 kb (hg19 chr20:43,029,924-43,060,029). At least 6 isoforms of HNF4alpha, generated by alternative promoter usage and C-terminal splicing, have been experimentally verified by more than one group (Fig. 5). Additional isoforms are still being identified. The second HNF4 gene in humans, HNF4gamma, is located on chromosome 8. Xenopus, but not mammals, have a third HNF4 gene, HNF4beta.
Figure 5. Structure of the human HNF4A gene and the isoforms generated by alternative splicing events.
Tissue Distribution and Role in vivo
HNF4alpha is expressed in liver (hepatocytes), kidney (proximal tubules), small intestine, colon and pancreas (beta cells), although hepatocytes remain one of the highest expressing cell types. There are also reports of HNF4alpha in the stomach, non beta cell compartments of the pancreas as well as the mammary epithelium, testes and ovarian cancer, although the role of HNF4alpha in those tissues has not been elucidated. HNF4alpha is expressed in many but not all human and rodent cell lines of hepatic, colonic and beta cell origin. The level of expression varies among the cell lines and isolates. Transformed cell lines of kidney origin, (e.g. , HEK293, Cos-7, Cos-1) do not express HNF4alpha. Primary hepatocytes and beta cells from human, mouse and rat also express HNF4alpha although the levels are highly dependent on the culture conditions. Expression profiles can be found at the Nuclear Receptor Atlas (NURSA).
Tissue-specific knockouts of the Hnf4a gene in mice have been created in liver, colon and pancreatic beta cells. The embryonic liver-specific knockout shows that HNF4alpha is required for the initial epithelial transformation of the fetal liver. The adult liver-specific knockout indicates that HNF4alpha is required for maintenance of the hepatic phenotype, including lipid homeostasis. The mice have a fatty liver and die within 6 weeks. Intestine/colon-specific knockouts in both the embryo and the adult verify that HNF4a is also required for the development and proper functioning of the gut; lower levels of HNF4alpha have also been noted in humans with inflammatory bowel (Crohn's) disease as well as a mouse model for experimental colitis. Two beta-cell-specific knockouts confirmed a role for HNF4alpha in regulating the insulin secretion pathway, although some discrepancies were noted. In contrast to humans, mice heterozygous for HNF4alpha do not show signs of diabetes. The reason for this is not known. See HNF4A, Transcription Factor Encyclopedia (TFe) for references.
HNF4alpha Target Genes
Since HNF4alpha was first identified as a factor that bound DNA response elements in the promoter region of liver-specific genes, many target genes were quickly identified for HNF4alpha in the liver using classical promoter deletion techniques. Those genes encode many of the proteins involved in intermediary metabolism, as well as those involved in blood maintenance, such as blood coagulation factors (F8, F9, F10, etc.). Point mutations in some of the blood coagulation gene promoters, as well as the HNF1A promoter, lead to human diseases such as hemophilia and diabetes, respectively. The human hepatitis B virus promoter/enhancer regions are also targets for HNF4alpha which is required for viral propagation. HNF4alpha is also a major regulator of xenobiotic and drug metabolism genes. Only a few HNF4alpha target genes in the other tissues (pancreas, kidney, small intestine, colon) have been identified by classical means. Recent studies using genome-wide techniques (such as Chip-chip and expression profiling) have identified more than a thousand additional potential targets of HNF4alpha although it remains to be determined how many of those are direct targets resulting from a specific HNF4-DNA interaction. More potential binding targets have been found by our latest in vitro study using protein binding microarray (PBM) and SVM. A list of targets can be found in (Bolotin E et al., 2010) and in HNF4A, Transcription Factor Encyclopedia (TFe).
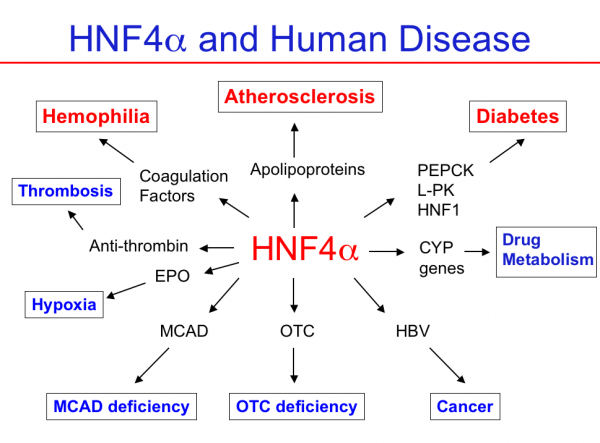
HNF4 and Human Disease
Mutations in the human HNF4A gene as well as in HNF4 binding sites have been linked directly to human diseases, most notably hemophilia and maturity onset diabetes of the young 1 (MODY1). Patients heterozygous for HNF4A exhbit macrosomia (high weight), hyperinsulinemia and hypoglycemia at birth, and then develop type 2 diabetes in young adulthood. Polymorphisms in the HNF4A gene as well as lower HNF4alpha mRNA and protein levels in colon have also been associated with Inflammatory Bowel and Crohn's Disease. Through other target genes, HNF4alpha is potentially linked to other diseases such as atherosclerosis and cancer. See HNF4A, Transcription Factor Encyclopedia (TFe) for details.
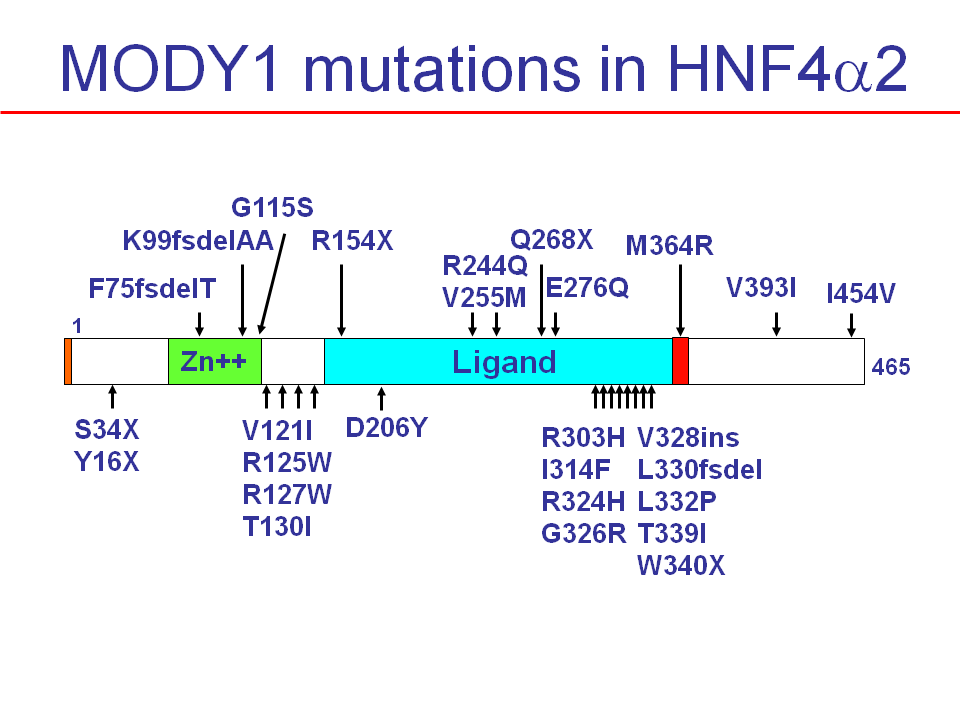
Figure 7. Shown are mutations found in the human HNF4alpha2 (NM_000457.3) coding sequence associated with Maturity Onset Diabetes of the Young 1 (MODY1). Given is the codon number and amino acid in single letter code. X, stop codon; fs, frameshift; del, deletion; ins, insertion. Not shown are mutations found in the HNF4alpha gene promoter associated with MODY1. Note, only heterozygous mutations have been identified.
Figure 8. HNF4a and human disease. HNF4a is linked to several human diseases via mutations in its binding sites (hemophilia, diabetes) or in the HNF4A gene itself (diabetes). It is also indirectly linked to many human diseases via the target genes it regulates.
For a more comprehensive and up-to-date review of HNF4, see Transcription Factor Encyclopedia (TFe).